A Guide to Balancing Chemical Equations.

Chemistry and when a student should take chemistry is currently a hot debate in education circles. There are several reasons this is happening:
- Increased biochemistry in intro biology courses
- Increased math requirements in high school
- The debate over whether chemistry is necessary at all.
Today we are going to focus on the math in Chemistry. Typically the bare minimum is Algebra II. This is because of the fact that Chemistry classes are very math-driven. Focused on formulas and proportions mostly.
Except for the role of factoring in balancing Chemical equations. Prior to balancing equations, you need to cover the basics of an atom (grab the parts of an atom worksheet) and the structure of the periodic table (printable periodic table).
Grab these resources by entering your information below.
What is balancing chemical equations?
Balancing chemical equations is about making sure that the same number of atoms of an element are on both sides of the equation.
Why is balancing equations important?
The law of conservation of matter says that matter can’t be created or destroyed. It only changes forms. That means the atoms on the reactants side must equal the number of atoms on the products side of the equation. While they are combined in different ways that the number of atoms of each element must be the same.
The parts of a chemical equation:
- subscript- the number of atoms of the element to make the compound or molecule ( DOES NOT CHANGE)
- coefficient- the number of molecules of the compound (this number can change – it WILL be the one to change when balancing equations)
- chemical formula- the actual compounds that are participants in the reactions. (DOES NOT CHANGE)
Looking for balancing chemical equations practice help or lesson ideas? Enter your details below to get the FREE worksheet sent to your inbox.
.
Steps to balance a chemical equation:
- Write down the equation
- Count the number of atoms of each element.
- Balance by adding coefficients.
- Balance hydrogen and oxygen last.
This is the same chemical equation from above BEFORE it was balanced:
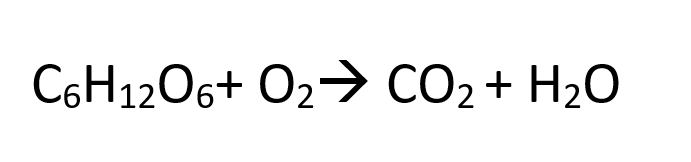
The only thing that can be changed in order to balance the equation is the coefficient (when coefficients are not present it is understood to be 1).
The coefficient is multiplied as if there were parentheses around the formula. It follows the distributive property.
Balance the equation by adding a coefficient
When deciding on the coefficient being able to find the lowest common multiple is essential. This will involve lowest common multiples and factoring.
Need a little reminder.
- The lowest common multiple is finding the lowest number that is a multiple of two or more numbers.
- Factoring is determining what are the numbers that could be multiplied to produce a number.
- Follow the rules for balancing equations. In this example, we will balance carbon first.
In the unbalanced equation, this is what we have:
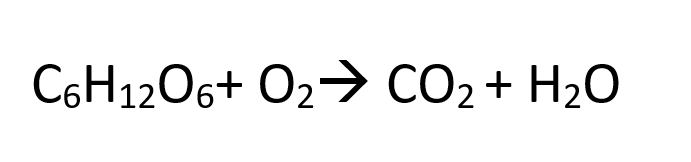

Balance Carbon First.
In order to balance carbon, we need to think of the lowest common multiple between the two numbers: 1,6. The lowest common multiple is 6. That will be the coefficient we add on the product side. When we add the coefficient it will multiply each atom (distributive factory).
The number of carbon atoms will increase to 6 and the number of oxygen atoms will be increased from 2 to 12 (6X2=12).


Now balance the hydrogen.
The lowest common multiple of 2 & 12 is 12. Using the coefficient of 6 (remember the distributive property). This will make 12 atoms of hydrogen on the product side.
It also changes the number of oxygen. Now there are 18 atoms of oxygen on the product side.


Now balance the oxygen
This one is tricky because there are two sets of oxygen on both sides of the equation. Since we have carbon and hydrogen balanced. The goal would be to only mess with oxygen if possible. In this case, it is, because oxygen is a reactant by itself.
Add a coefficient of 6 changes the atoms of oxygen from 2 to 12. Adding the 12 to the 6 in glucose gives us the magic number of 18.


Looking for ways to practice balancing chemical equations?








One Comment
Comments are closed.